Joffe and colleagues found that 16% of the orphan drugs approved from 2009 to 2015 were for subsets of common diseases defined by biomarkers. Given that the Orphan Drug Act was supposed to incentivize the development of drugs for low-prevalence conditions, the spirit of the law is not quite being followed if orphan drugs are being used by large numbers of patients because the drugs have multiple indications or are being used off-label, says Joffe. Pushback from insurers Insurers are beginning to push back some—not a lot, but some—on paying for orphan drugs with big price tags. In May, Harvard Pilgrim Health Care signed a contract with Amgen for its PCSK9 inhibitor, an LDL cholesterol lowering drug called Repatha.
Under this contract Amgen will pay HPHC and its members a full refund if a member requires hospitalization for a heart attack or stroke after taking Repatha for at least six months and achieving an appropriate level of compliance. Designated an orphan drug in 2013, Repatha has market-exclusivity until 2022. Last year, Anthem said that it would not cover Exondys 51, the controversial drug for Duchenne muscular dystrophy priced at $300,000 per patient per year. Despite FDA approval, Anthem said Exondys 51 was investigational and not medically necessary. FiercePharma reported that UnitedHealthcare would cover the drug, while Aetna and Express Scripts would review the clinical data before deciding on coverage.
Payers also are pushing back on Spinraza, the new cancer, and so on. “The current system of classifying by both organ of origin and molecular identity is creating a situation wherein almost every medication can be an orphan,” they wrote. Pricing, profit, and the possibility of a blockbuster therapy are among the reasons the orphan drug market has become so attractive for the nation’s largest pharmaceutical companies, notes Enrique Seoane Vazquez, an associate professor and director of the International Center for Pharmaceutical Economics and Policy at the Massachusetts College of Pharmacy and Health Sciences. Seoane Vazquez is one of the authors of an article published earlier this year in the Orphanet Journal of Rare Diseases on how to reconcile economic incentives and meet patients’ needs.
When the orphan drug legislation was passed, small pharmaceutical companies and universities were developing medications for rare diseases, Seoane Vazquez says. “Right now, all the largest pharmaceutical companies are involved in the research and development of orphan drugs.” Along with the change from small companies to larger ones, orphan drugs have become a major cost concern for affected patients, in part because of the advent of so many high-deductible health plans. For these and other reasons, the orphan drug market needs more scrutiny from regulators and those with the power of the purse in American health, meaning private and public payers, says Steven Joffe, MD, an associate professor at the University of Pennsylvania Perelman School of Medicine and coauthor of an article published earlier this year on the biomarkerdefined subsets of diseases.
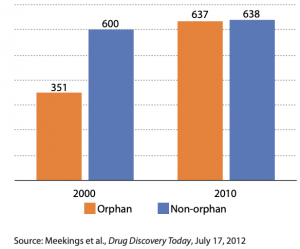
Increasing value of orphan drugs
Average expected lifetime revenue from orphan drugs vs. non-orphan drugs, adjusted for inflation, 2000 vs. 2010 US $millions

Paul Lendner ist ein praktizierender Experte im Bereich Gesundheit, Medizin und Fitness. Er schreibt bereits seit über 5 Jahren für das Managed Care Mag. Mit seinen Artikeln, die einen einzigartigen Expertenstatus nachweisen, liefert er unseren Lesern nicht nur Mehrwert, sondern auch Hilfestellung bei ihren Problemen.
